Join us as we embark on a journey into the future of drug safety monitoring!
The Hyve is thrilled to announce the upcoming webinar series "Beyond Traditional Approaches in Drug Safety: The Power of Semantic Modeling and Knowledge Graphs", aiming to explore the application of semantic technologies in drug safety monitoring.
In the ever-evolving world of medicine, the practice of monitoring and evaluating the safety of pharmaceutical products (pharmacovigilance) is critical to ensure patient safety. However, the amount of complex and multi-source data available creates numerous challenges. Also, the task of accurately detecting the causal relationships between adverse events and medications remains a pivotal concern. Responding to these challenges, The Hyve is organizing a series of webinars focused on novel solutions to support and advance data-driven processes in pharmacovigilance.
Semantic Models and Knowledge Graphs: The Future of Drug Safety
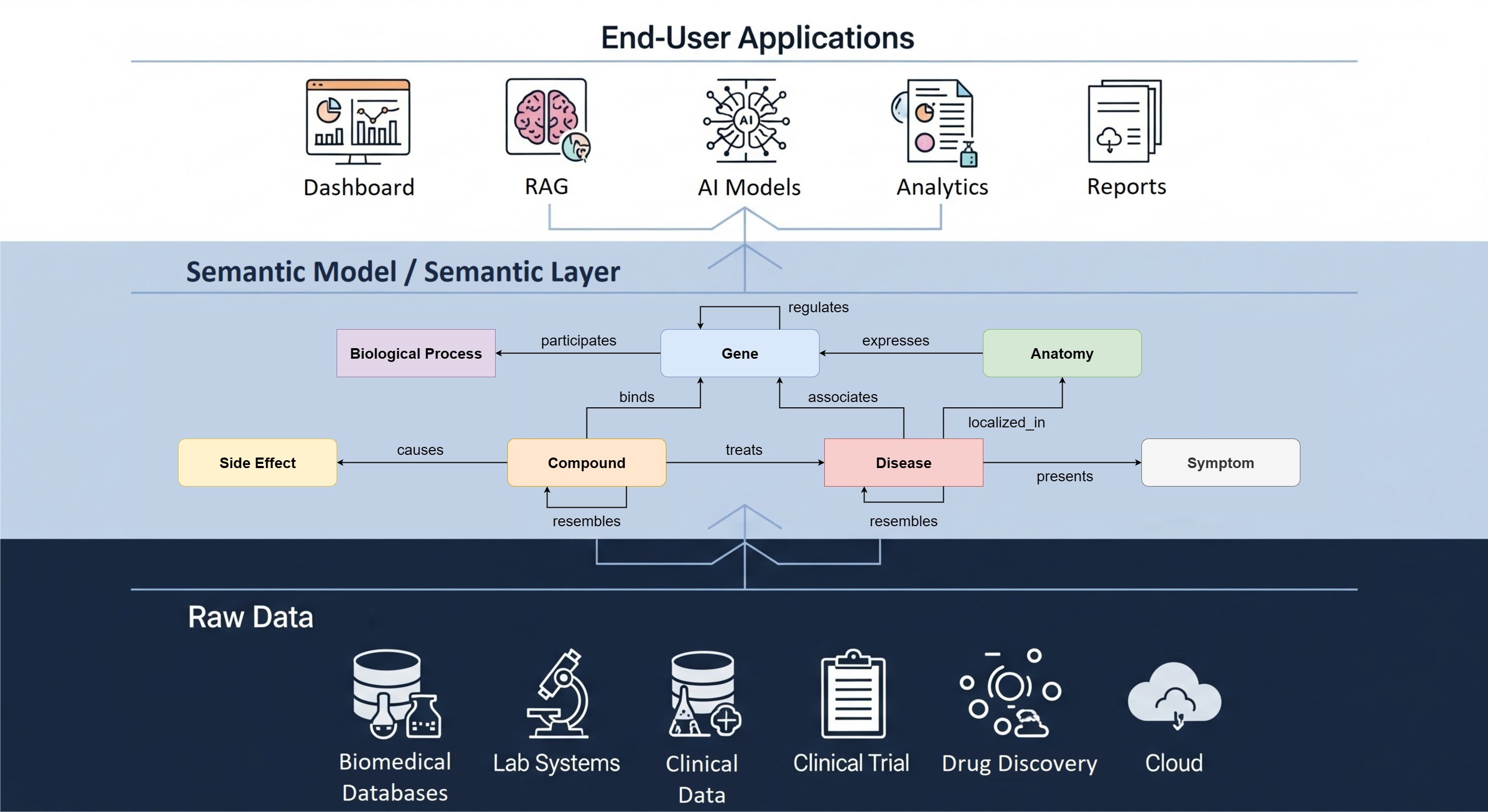
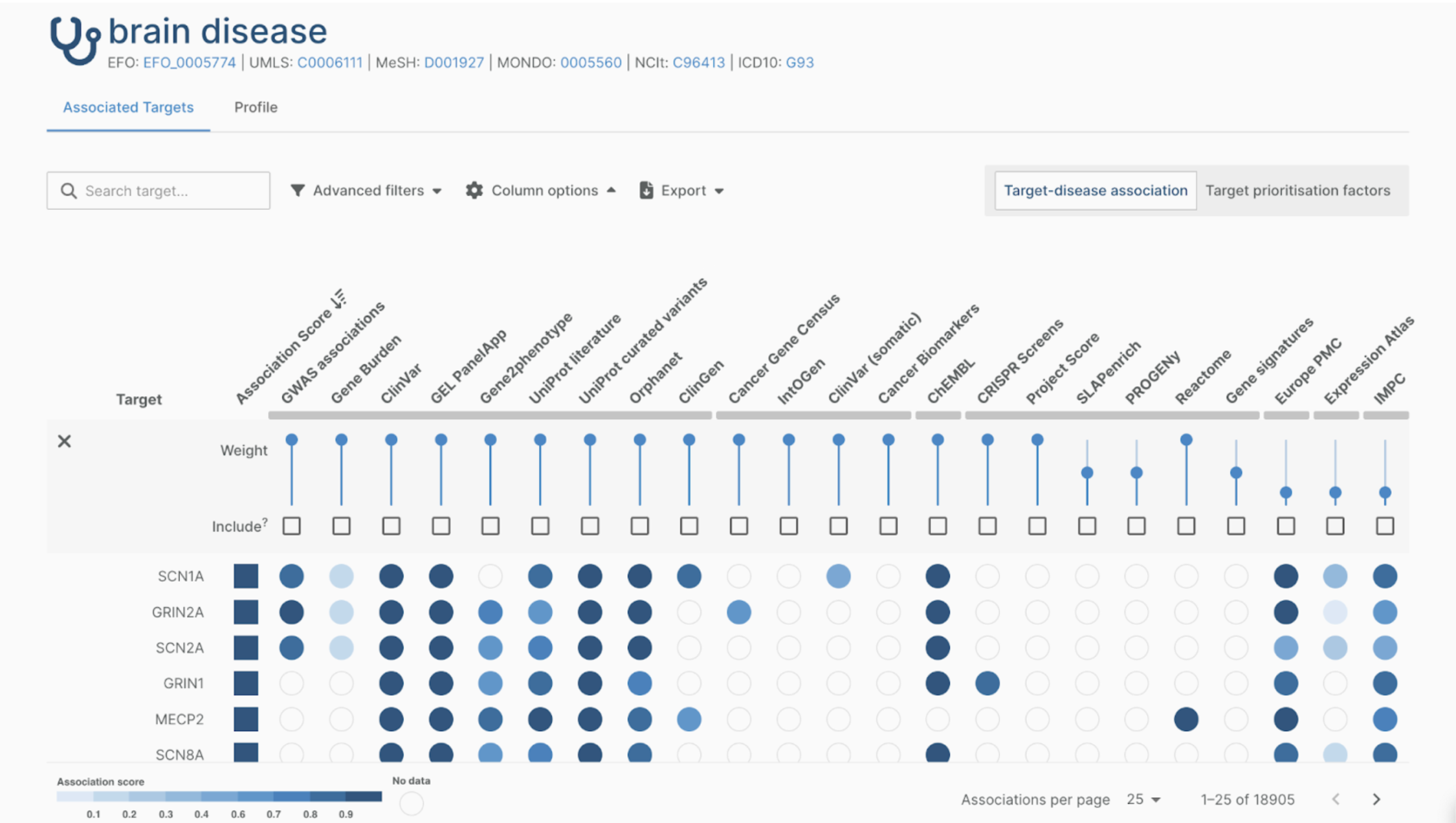
At the heart of this series lies the application of Semantic Modeling and Knowledge Graph technologies to detect, assess, understand and monitor drug safety issues. By offering a paradigm shift in how data and relationships are represented, these technologies provide a structured, context-rich, scalable, and machine-understandable output. Our webinar series seeks to delve into the transformative capabilities of these technologies in the field of pharmacovigilance.
Highlights of the Webinar Series
Mark your calendars on 10th October 2023, from 15.30 to 16.30 CEST for the debut session "Integrating Biomedical Ontologies with OMOP: Opportunities for Drug Safety Monitoring". During this first webinar, we will explore the convergence of data harmonization and semantic models in the context of drug safety monitoring. Dr. Tiffany Callahan (Postdoctoral Research Scientist, IBM Research Almaden) will present a 30-minute talk on the topic, focusing on "Ontologizing Health Systems to Facilitate Translational Discovery and Improve Drug Safety."
The subsequent panel discussion will feature experts in the field, including Dr. Tiffany Callahan, Sofia Bazakou from The Hyve's Real World Data team, and Elpida Kontsioti from The Hyve’s Research Data Management team. Key themes to be explored include the potential of semantic models in enhancing the identification of adverse events, the challenges and opportunities in harmonizing diverse datasets, and the OHDSI community's initiatives driving data integration and sharing for drug safety monitoring applications.
Your Invitation Awaits: Register Now!
This informative webinar series will shed light on innovative methodologies in data harmonization, semantic modeling, and knowledge representation that are reshaping the landscape of pharmacovigilance. The first webinar is a unique opportunity to gain insights from experts and be part of a transformative conversation around the future of drug safety.
Register for the first webinar
Stay tuned for the second webinar
The Hyve is also excited to announce that the second webinar in the series, focusing on "Knowledge Graphs for Multi-Source Data Analysis in Pharmacovigilance," is tentatively scheduled for November 14, 2023. Be sure to subscribe to our newsletter to receive the latest updates!