The Hyve is happy to announce the public release of the EHDEN Portal, the tool through which the EHDEN framework can be accessed. The portal was officially launched at the OHDSI Europe Symposium 2022, held June 24-26 in Rotterdam, the Netherlands. The EHDEN Portal is based on a content management system that allows linkage to a range of underlying applications. Julia Kurps, Real World Data Team Lead at The Hyve and Co-Lead of the technical Work Package EHDEN, described the ins and outs of the new portal in a presentation titled "Findable, standardized data at scale through the EHDEN Database Catalogue." Watch her presentation below.
EHDEN Portal and Data Catalogue Launch
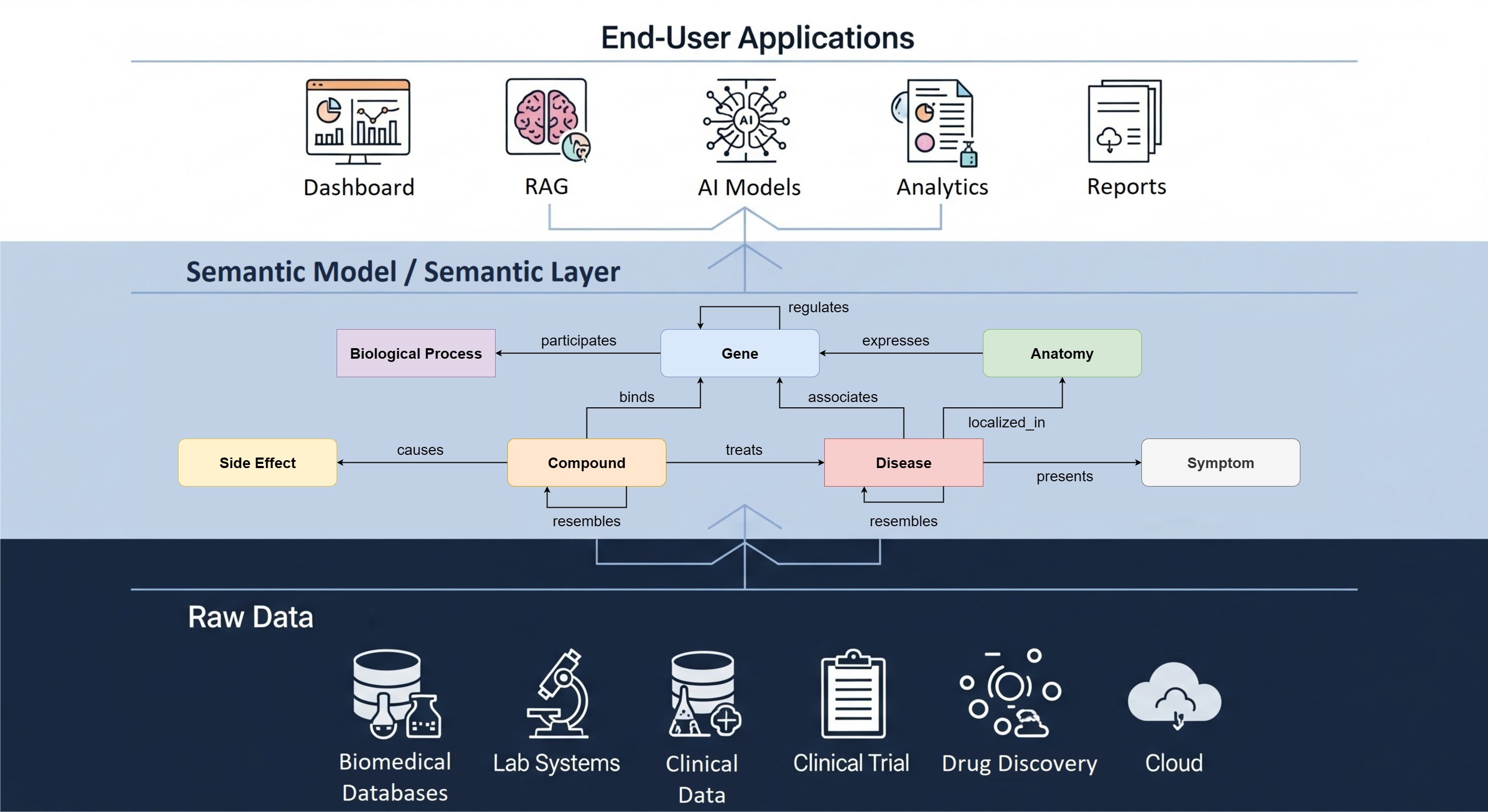
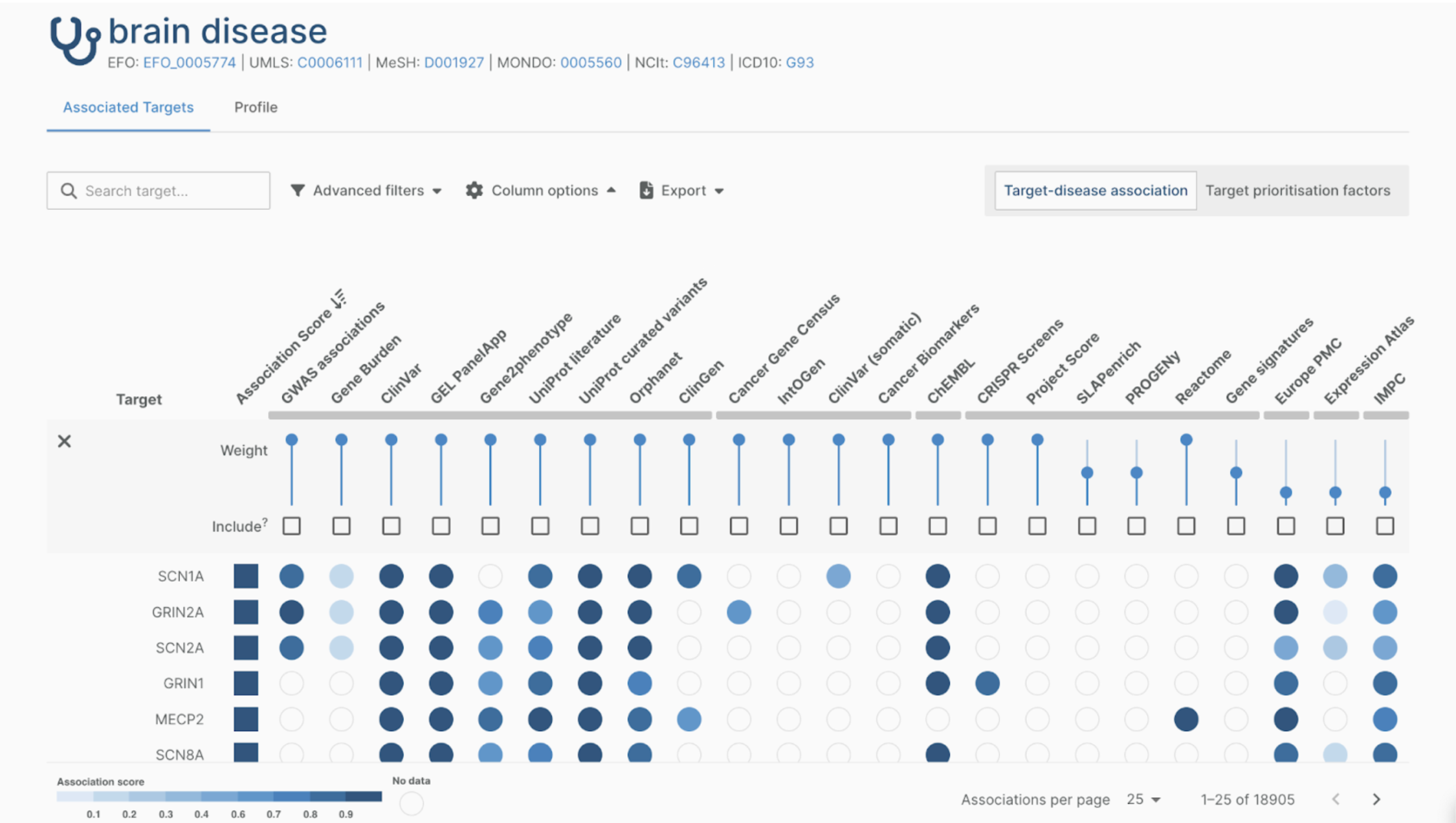
The EHDEN Portal includes a Data Partner Catalogue and Feasibility Dashboards that support data discoverability. Such tools are essential for addressing the challenge that everyone working with observational health data faces: having ‘a data ocean but a desert for analysis’.
Currently, 67 EHDEN Data Partners are included in the Catalogue. These partners either provide metadata on European observational health data in the OMOP format or convert their source data to the OMOP common data model (CDM). All Data Partners who receive IMI EHDEN funding will add their metadata to the Catalogue, resulting in further network growth in the coming months and years.

While the Catalogue contains metadata, the Feasibility Dashboard gives access to the database and allows users to explore the data based on characteristics such as geographical spread, demographics, data domains, as well as information about data provenance, observation periods, hospital visits, and condition occurrence.

This Portal demonstrates how EHDEN's mission is becoming a reality: to provide a new paradigm for the discovery and analysis of observational health data in Europe by building a large-scale, federated network of data sources standardized to a common data model (OMOP). The Portal supports collaborative research that will translate into better insights into disease progression and treatment results. This will eventually enable improved medical decision-making and better treatments for patients based on real-world evidence.
About EHDEN
EHDEN (European Health Data and Evidence Network) is a federated data network designed to address the challenges researchers face in the generation of real-world evidence (RWE) from real-world data (RWD). EHDEN aims to maximize the transformational potential of RWD and RWE at scale to facilitate standardized and faster observational research that improves insights into disease, treatment, and outcomes for patients across Europe.
Read more about EHDEN’s presence at the OHDSI Europe Symposium 2022, including reflections on the Portal’s launch by esteemed community members, here.
Funding Statement
The European Health Data & Evidence Network (EHDEN) has received funding from the Innovative Medicines Initiative 2 Joint Undertaking (JU) under grant agreement No 806968. The JU receives support from the European Union’s Horizon 2020 research and innovation program and the European Federation of Pharmaceutical Industries and Associations (EFPIA).