One of the aims of digital health is to improve patient care and to offer a better healthcare experience to patients. Also in recent years, this rapidly advancing field has been providing great opportunities to radically transform the way clinical and research studies are conducted. Since 2016, when the RADAR-base platform was designed and launched, The Hyve has noticed that digital studies are moving from a purely observational approach based on systematic data collection to a more interventional design based on participant feedback. In other words: there is an ongoing shift from the traditional site-centric to a patient-centric paradigm. At the Hyve we have witnessed this change in the work we did in collaboration with University College London (UCL), King’s College London (KCL), the IMI H2O consortium and the DynaMORE project. The ongoing shift is a clear sign that times are changing: participants are more informed and have a better understanding of the value of their data. Also, The Hyve chose the open-source development and licensing model as it enables the design of more and new patient-centric digital interventions.
Developing patient-centric digital interventions with RADAR-base

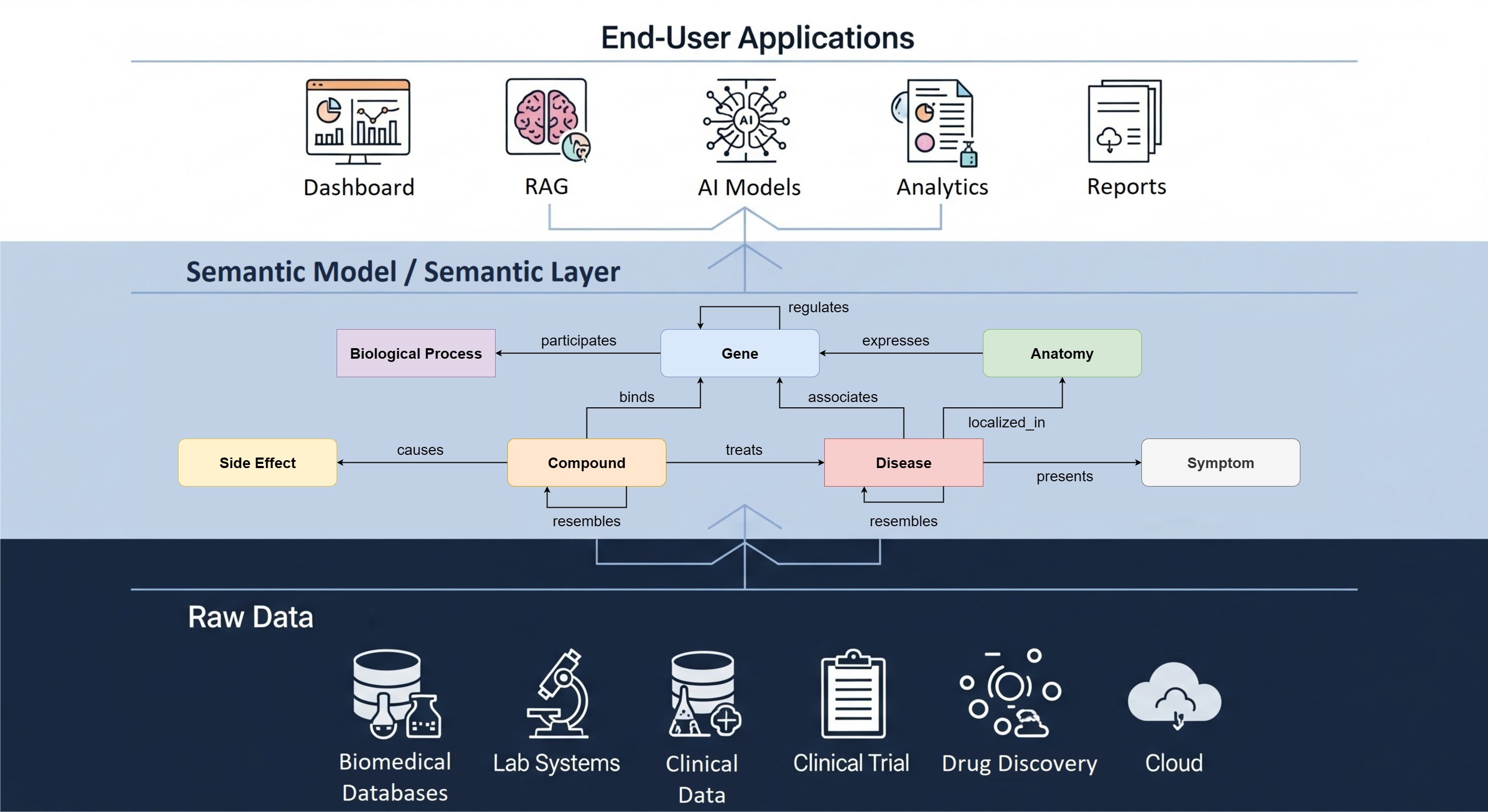
The RADAR-base platform has been adopted in several grant-based studies for remote patient assessment in various fields: neuro-degenerative diseases, mental health disorders, diabetes, pediatric diseases and in studies aimed to evaluate the quality of life. When The Hyve started designing RADAR-base in 2016 for the IMI RADAR-CNS consortium, the goal was to support the work of medical and pharmaceutical researchers and build a platform that could be widely adopted, without vendor lock-in. We wanted to create a solution that could be easily used in a range of digital and remote studies, leveraging participant generated data from a variety of wearable devices, activity trackers and smartphones in conjunction with medical grade equipment. We focused on building a platform to collect standardized, high quality data. In fact, RADAR-base enables near real-time data streams so that researchers can continuously monitor compliance of participants and intervene quickly if data collection or transmission is interrupted to prevent data loss. RADAR-base helps scientists make sense of data coming from various sources, in multiple formats, at different timepoints/over different time spans.
Active participation in the roadmap of the platform
While our focus is on developing high-quality code, our core customer base, the researchers, try to find ways to increase participant compliance. For research studies to generate relevant outcomes for patients, researchers and participants have to work together when shaping the study design and a lot of time is devoted to discussing incentives for study participation and adherence. Participants want to be engaged and researchers need to have a clear picture of the incentives for their envisioned study participants.
The shift towards more patient-centric solutions is a relatively recent trend in the IT market. IMI RADAR-CNS, and its successor IMI RADAR-AD, started with patients in mind and involved a patient advisory board from day one of the project proposal creation. In fact, collection of patient feedback has been a regular part of software development. The RADAR-base platform has been, and continues to be built, based on the feedback of patients, their families and patient organizations.
Access to aggregate patient information
- By collecting and harmonizing raw data from study participants, directly from mobile apps and sensors as well as from wearable devices, RADAR-base enables scientists to perform their own data analyses and obtain personalized insights for every participant.
- Only pseudonymised data is collected in the RADAR-base platform.
- Only the researchers that manage a study have control over who has access to the data, The Hyve does not. The platform manager is the data custodian, not the software provider.
- Participants receive tailored questionnaires and notifications - the digital intervention - right on their phone.
- The monitoring mobile app is unintrusive; it just runs in the background without additional burden for the enrolled subject or draining the batteries.
- In most use cases, patients do not have any interaction with the platform except via the apps. Participants don't have to do anything else besides installing the applications on their smartphone.
- Patient data is protected and access is restricted.
The Hyve remains focused on inventing ways to make data from different sources more FAIR, and therefore more valuable for medical research. There is a need in the market for alternative business models that protect research data in general and patient data in particular. For instance, the choice for a specific remote monitoring device should not only be driven by “data quality” but also be supported by a well-informed choice of the patient. The participant should always be aware and completely onboard with the way their data is collected, handled and used.
The RADAR-Engage testimonial, Katie White

“I have been working as a research assistant in the RADAR-CNS consortium for over three years now, recruiting and following-up participants who have been providing data through the RADAR-base platform as part of the major depressive disorder branch of the study. I am particularly interested in why and how people with depression choose to engage with wearable devices, activity trackers and smartphones to track their symptoms in a research context, and I began a PhD last year to explore this further. Based on qualitative work with participants, I designed some new in-app components to be delivered via the user-facing section of RADAR-base, the questionnaire app, which I hope will promote objective and subjective engagement with symptom tracking remotely. I approached The Hyve to develop the ‘progress visualisation’ aspect of the work − a dynamic graph that visualises weekly completion rates of tasks in real time. The whole journey from conception to completion was extremely efficient and well-managed, and I worked with a software engineer who was able to produce exactly what I had envisioned in a very quick turnaround. I am currently running a 12-week randomized controlled trial with the adapted questionnaire app which, providing it is successful, should optimise the use of the RADAR-base system and remote measurement technologies for maximum data collection (whilst maintaining minimum user burden) in further mental health research studies, clinical trials and clinical practice."
Funding statement and Disclaimer
The RADAR-base platform was developed under projects IMI RADAR-CNS, IMI RADAR-AD, Bigdata@Heart and IMI H2O which received funding from the Innovative Medicines Initiative 2 Joint Undertaking under grant agreement numbers 115902, 806999, 116074 and 945345 respectively. This Joint Undertaking receives support from the European Union’s Horizon 2020 research and innovation programme and EFPIA. See www.imi.europa.eu for more details.
Neither IMI nor the European Union and EFPIA are liable for any use that may be made of the information contained on this website.